Decoding Sleep: What Your Body Does At Night?
A deep dive into the science of sleep: the brain architecture that drives it, the expert consensus on how to measure its quality, and its role as a window into systemic health.
What is sleep?
TL;DR: Far from being a state of “nothingness,” sleep is a highly active, neurobiological process of “cellular housekeeping” that restores the brain and the body.
I used to be the person who dreamt of functioning perfectly on five or six hours of sleep. I saw sleep as a tax I had to pay on my productivity. Years of neuroscience training changed that; I am now someone who protects an eight-hour window, knowing it is the single most effective thing I can do for my health.
We spend about one-third of our lives asleep, yet we are only beginning to unlock its complexity. For a long time, we viewed sleep as a passive state - a “light switch” that simply turned the brain off. In reality, sleep is a masterpiece of biological engineering. While you drift off, your brain flushes out metabolic waste through the glymphatic system, repairs damaged tissues, and processes memories and emotional data. From a scientific point of view, sleep is defined as a bio-behavioral state observable through changes in brain electrical activity, altered consciousness, reduced sensory responsiveness, and decreased muscle tone [1].
What does your brain do when you sleep?
TL;DR: Sleep is orchestrated by a specialized network of “switches” in the hypothalamus and brainstem, coordinated by a master clock that syncs our biology with the sun.
For neuroscience aficionados like myself, understanding sleep requires a look at the “hardware” behind the behavior. This hardware is your brain’s anatomy and it determines how you sleep.
The Hypothalamus: The Master Clock
The hypothalamus is a peanut-sized structure that acts as the brain’s primary command center for homeostasis. Within this structure sits a tiny cluster of neurons called the Suprachiasmatic Nucleus (SCN). Think of the SCN as your body’s master clock. It receives light information directly from the retina in your eyes. When the sun goes down and light fades, the SCN signals the rest of the brain to shift from “active mode” to “recovery mode.” It is the SCN that dictates your circadian rhythm - the 24-hour cycle that tells you when it’s time to sleep.
The Pineal Gland: The Starting Pistol
While the SCN is the clock, the Pineal Gland is the messenger. Once it receives the signal cascading from the SCN, the pineal gland begins producing melatonin. It’s a common misconception that melatonin is a sedative that knocks you out. In reality, it acts more like a biological starting pistol, signaling to the rest of your systems that the race for sleep has officially begun [2].
The Brainstem: Quieting the Brain
In our last article, we talked about the Autonomic Nervous System and its two branches: the Accelerator (Sympathetic) and the Brake (Parasympathetic). As you prepare for sleep, the brainstem withdraws the ‘Accelerator’ signals - the wake-promoting neurotransmitters that kept you alert and responsive. In parallel, the hypothalamus releases GABA, the brain’s primary inhibitory neurotransmitter, actively quieting the brain’s arousal centres. Together, this essentially ‘muffles’ sensory input from the outside world, allowing you to drift off. During REM sleep, the brainstem sends a temporary “disconnect” signal to your muscles, ensuring you don’t physically act out your dreams - a vital safety feature of our biological architecture.
What does a night of sleep look like?
TL;DR: Sleep is not a uniform state but a series of 90-minute cycles composed of REM and non-REM stages, each serving a distinct purpose for physical and mental recovery.
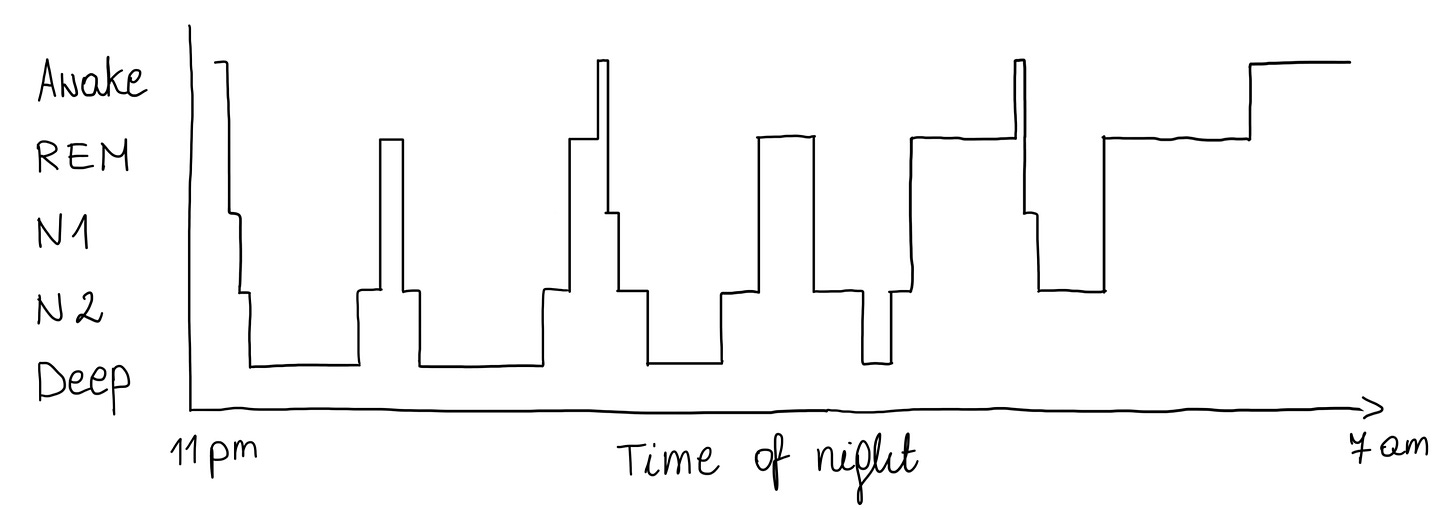
After learning how your brain regulates sleep, let’s talk about what sleep is composed of. When you look at your wearable data, you’ll see your night broken down into a “hypnogram” - a map of your sleep stages. These stages are defined by specific patterns of brain wave activity and are broadly divided into REM (Rapid Eye Movement) and Non-REM (NREM) sleep.
Fig. What a typical night of sleep might look like.
Think of a night of sleep like a healthy meal: you need every micronutrient, to feel “full” the next morning.
Non-REM Stage 1 and 2: Light sleep (~50% of total sleep)
Stage 1: This is the “dozing off” phase. Your brain waves begin to slow, but you’re still easily awoken.
Stage 2: This makes up the bulk of your night. Your heart rate drops, and your brain produces “sleep spindles” - short bursts of activity that are thought to be essential for memory consolidation.
Non-REM Stage 3: Deep Sleep (13-23% of total sleep)
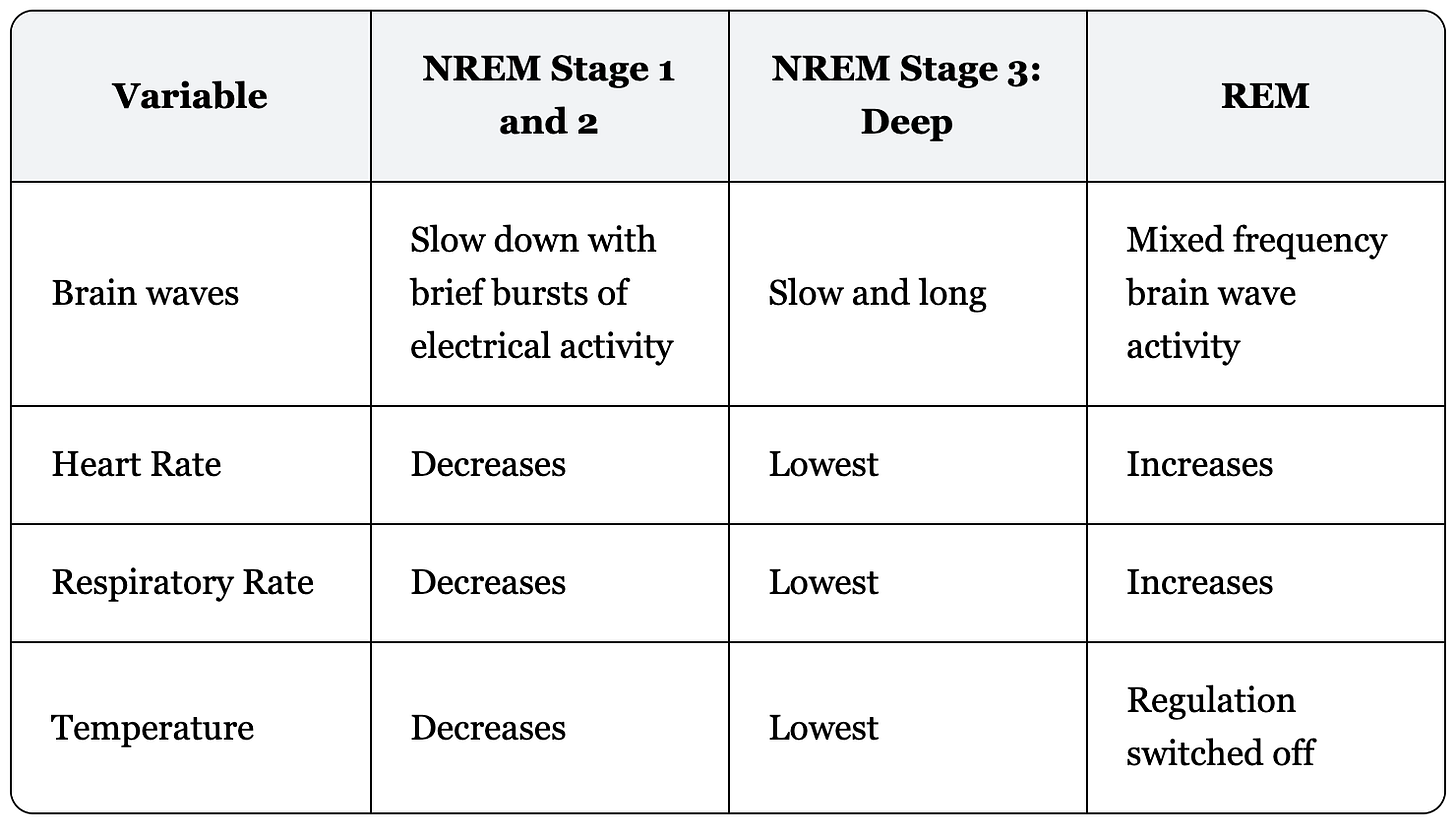
Often called “Slow Wave Sleep,” this is the most restorative stage for the body. During this phase, your brain produces large, rhythmic waves, blood flow is diverted from the brain to the muscles to repair tissue, the immune system is boosted, and growth hormones are released. If you wake up feeling physically “heavy” or groggy, you were likely pulled out of this deep state.
REM Sleep (20-25% of total sleep)
While NREM is for the body, REM is for the mind. During REM, your brain activity looks remarkably similar to when you are awake. Your eyes move rapidly behind your lids, and your body enters a state of temporary paralysis to prevent you from acting out your dreams. This is where we process emotions and integrate complex memories. A lack of REM sleep often leaves us feeling “foggy” on the next day.
One thing worth knowing is that these stages are not evenly distributed across the night. Deep sleep dominates the first half - your brain prioritises physical restoration early on. REM, on the other hand, becomes increasingly concentrated in the second half, particularly in the final one to two hours before you wake. This is why sleep timing matters as much as sleep duration. An early alarm doesn’t just shorten your night - it disproportionately cuts your REM.
Changes in physiological variables during different sleep stages.
How much should you sleep?
TL;DR: While the expert consensus for healthy adults is 7–9 hours, the ideal duration is a dynamic target that shifts across the lifespan.
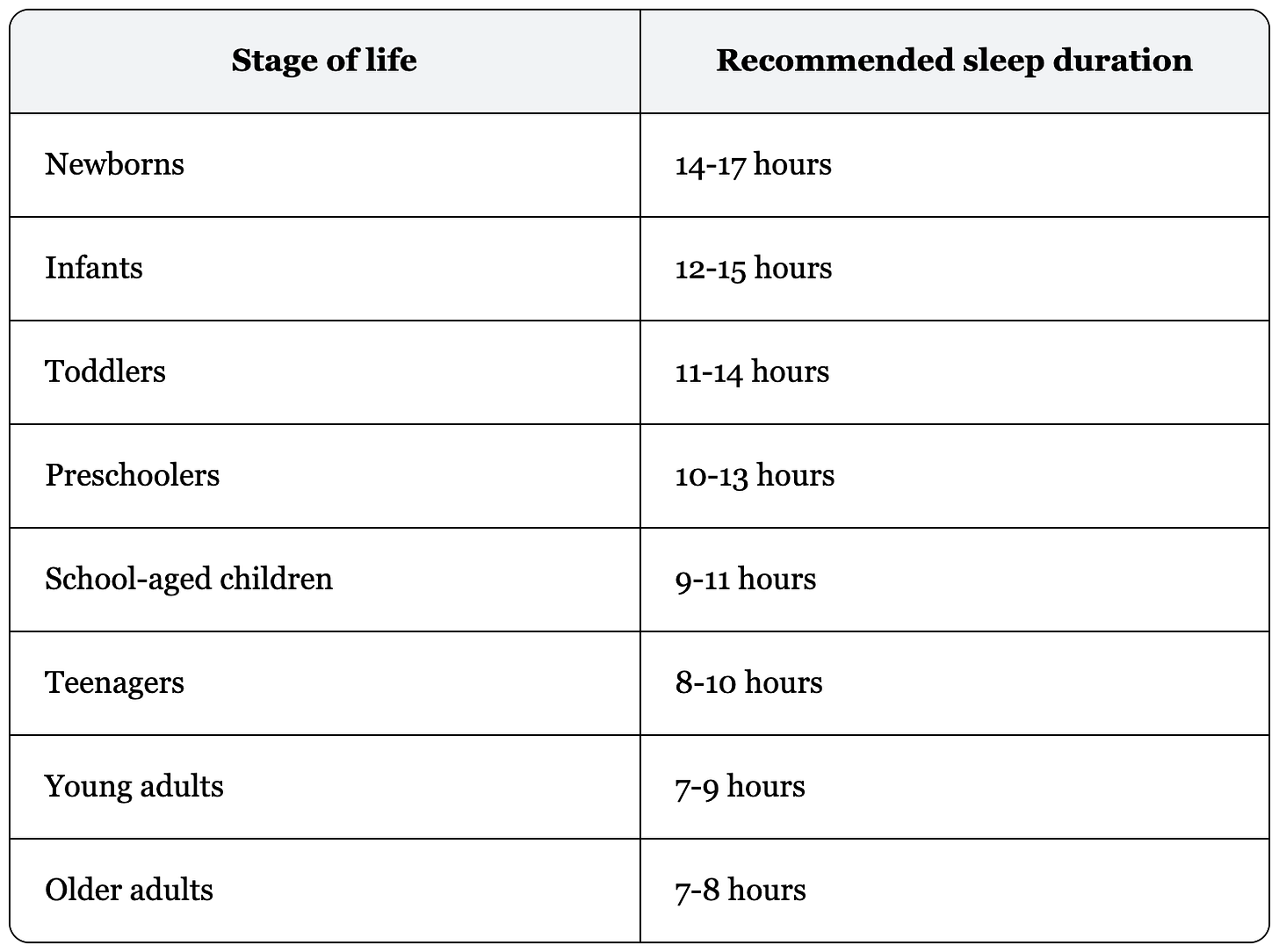
In 2015, the National Sleep Foundation (NSF) conducted a comprehensive review to establish guidelines for sleep duration [3]. The most significant finding is that our sleep requirements are not “one size fits all” - they shift significantly as our brain and body age. While newborns require nearly double the sleep of an adult to support rapid development, the biological requirement for adults stabilizes between 7 and 8-9 hours.
For the majority of adults, 7 hours is the biological “floor.” When we consistently get less than this, we begin to see a measurable impact on our health data. Specifically, short sleep duration is a primary driver of sympathetic dominance - meaning if you don’t hit these duration targets, the “Accelerator” stays active, your heart rate remains elevated, and your HRV baseline will likely drop over time.
What defines sleep quality?
TL;DR: Sleep quality is measured by “continuity” - how efficiently you stay asleep and how quickly you return to sleep if interrupted.
The duration of sleep, however, doesn’t tell you the whole story. In 2017, an expert panel from the NSF performed a systematic review of 277 studies combined with voting to define exactly what “good sleep quality” looks like across life-span [4]. They concluded that quality is best indicated by sleep continuity variables. These are the metrics that determine whether your sleep is consolidated or fragmented. According to the NSF panel, for a healthy adult, “good quality” sleep typically meets the following criteria:
Sleep Latency: You should be falling asleep within 30 minutes of getting into bed. If you’re lying there for an hour running through tomorrow’s to-do list, that’s a signal worth paying attention to.
Awakening: Waking once in the night is normal. Waking two, three, or four times, for more than 5min, is a sign of fragmented sleep that adds up over time.
Waking After Sleep Onset (WASO): If you do wake up after falling asleep, staying awake for more than 20 minutes overall will also affect your sleep.
Sleep Efficiency: This is the ratio of time actually asleep versus time spent in bed. The target is 85% or above. If you’re in bed for eight hours but only sleeping six, your efficiency is 75% - and your body knows the difference, even if you don’t.
How does your wearable track sleep?
TL;DR: While clinical sleep studies measure brain activity directly, wearables use movement and heart rate as “proxies” to estimate your sleep stages.
It is important to understand that how sleep is measured differs significantly between clinical laboratory conditions and the wearable world. Distinguishing between these two methods helps explain why your watch might miss a wake-up event or miscalculate a specific stage.
The Gold Standard: Polysomnography (PSG)
In clinical settings, sleep is measured using Polysomnography. If you were to participate in a sleep study, you would be hooked up to a variety of sensors that track:
Brain Waves (EEG): The only direct way to see which sleep stage the brain is in.
Eye Movements (EOG): Essential for identifying the “Rapid Eye Movement” in REM sleep.
Muscle Tone (EMG): Used to detect the physical paralysis that occurs during REM.
Heart Rate and Respiration: To monitor autonomic health and breathing disorders.
The Wearable Approach: Actigraphy and Photoplethysmography (PPG)
Because we cannot easily measure brain waves at home, wearables rely on two primary sensors to infer when and how we sleep:
Actigraphy (Accelerometers): This measures your movement. The logic is simple: if you are moving, you are likely awake; if you are perfectly still, you are likely asleep.
Photoplethysmography (PPG): It measures your heart rate, HRV and respiratory rate.
Accuracy and Validation
Researchers use validation studies to compare wearable data against Polysomnography. These studies show that while wearables are good at tracking total sleep duration and “wake vs. sleep” transitions, they are less precise at distinguishing between specific stages like light vs. REM. I will discuss wearable accuracy in sleep in my next article, stay tuned!
Why is sleep so important?
TL;DR: Sleep disturbances are clinical predictors for cardiovascular diseases, metabolic dysfunction, mental health disorders and long-term cognitive decline.
When we think of sleep, we often associate it with productivity and performance. In reality, it is much more than this. Sleep disturbances are linked to poor cardiovascular health, diabetes, mental health, and even dementia.
Cardiovascular Health: A meta-analysis of over 400,000 participants found that both short and long sleep durations were associated with an increased risk of stroke and coronary heart disease [5]. The evidence is so compelling that in 2022, the American Heart Association officially added sleep duration to its “Life’s Essential 8”- the checklist of the most important predictors of cardiovascular health - placing it alongside blood pressure, cholesterol, and blood sugar [6].
Metabolic Regulation: A 2015 meta-analysis described a “U-shaped” risk profile for diabetes: the risk is lowest at 7–8 hours, while both shorter and longer sleep durations significantly increase the likelihood of developing Type 2 Diabetes [7].
Dementia: A study published in Nature Communications found that consistent sleep of 6 hours or less in middle age is associated with a 30% increase in dementia risk, independent of sociodemographic, cardiometabolic, or mental health factors [8].
Mental Health: I recently attended back-to-back lectures by Prof. Terrie Moffitt and Prof. Avshalom Caspi, two of the world’s leading researchers in longevity and human development. What struck me most was their discussion of a recent publication in Nature Medicine where researchers identified sleep disturbances in adolescents as a robust predictor of psychiatric illness - surpassing even adverse childhood experiences and family mental health history as a predictive tool [9].
These are just a few examples, and for many of them, we are still uncovering the exact biological mechanisms. However, they demonstrate how powerful sleep is for healthy development, living, and aging.
Lessons learned: Healthy sleep is the best health outcome
Although we are still uncovering the exact biological mechanisms behind all these results, one thing is clear: sleep emerges as a promising therapeutic and preventative approach for health across different domains. This is fundamentally good news. Why? Because for many people, sleep is a target we can actively support. Sleep therapies, such as Cognitive Behavioral Therapy for Insomnia (CBT-I), already exist and have been proven effective in helping people navigate and improve their sleep patterns [10]. This is particularly exciting for the field of mental health, where traditional therapies often fail.
I strongly believe that wearables will play a vital role in this shift. They give us a window into our patterns over a longer period of time - something that was never achievable on this scale before. This doesn’t mean you should obsess over a single sleep score recorded by your watch or ring. It means you can observe how your sleep looks over weeks and months, adjust your environment where possible, and, most importantly, have the longitudinal data to seek professional help if you see persistent disturbances. Your wearable isn’t just a tracker; it’s a tool for advocacy and early intervention.
If this was worth your time, subscribe for more — each post explores the science behind wearable health data: how it’s measured, what it means, and what the research says.
This post is part of The Science Behind Wearables - a series explaining the health metrics your devices track, built around the health scores we’re developing with the Open Wearables team. Open Wearables is an open-source platform for standardized access to health data from consumer wearables, supported and maintained by Momentum.
Know someone obsessed with wearables? Forward this their way.
Sources:
[1] Luyster, F.S., Strollo, P.J., Zee, P.C. and Walsh, J.K. (2012). Sleep: A Health Imperative. Sleep, 35(6), pp.727–734. doi:https://doi.org/10.5665/sleep.1846.
[2] Arendt, J. and Skene, D.J. (2005). Melatonin as a chronobiotic. Sleep Medicine Reviews, 9(1), pp.25–39. doi:https://doi.org/10.1016/j.smrv.2004.05.002.
[3] Hirshkowitz, M., Whiton, K., Albert, S.M., Alessi, C., Bruni, O., DonCarlos, L., Hazen, N., Herman, J., Adams Hillard, P.J., Katz, E.S., Kheirandish-Gozal, L., Neubauer, D.N., O’Donnell, A.E., Ohayon, M., Peever, J., Rawding, R., Sachdeva, R.C., Setters, B., Vitiello, M.V. and Ware, J.C. (2015). National Sleep Foundation’s updated sleep duration recommendations: final report. Sleep Health, 1(4), pp.233–243. doi:https://doi.org/10.1016/j.sleh.2015.10.004
[4] Ohayon, M., Wickwire, E.M., Hirshkowitz, M., Albert, S.M., Avidan, A., Daly, F.J., Dauvilliers, Y., Ferri, R., Fung, C., Gozal, D., Hazen, N., Krystal, A., Lichstein, K., Mallampalli, M., Plazzi, G., Rawding, R., Scheer, F.A., Somers, V. and Vitiello, M.V. (2017). National Sleep Foundation’s sleep quality recommendations: first report. Sleep Health, 3(1), pp.6–19. doi:https://doi.org/10.1016/j.sleh.2016.11.006.
[5] Cappuccio, F.P., Cooper, D., D’Elia, L., Strazzullo, P. and Miller, M.A. (2011). Sleep duration predicts cardiovascular outcomes: a systematic review and meta-analysis of prospective studies. European Heart Journal, 32(12), pp.1484–1492. doi:https://doi.org/10.1093/eurheartj/ehr007.
[6] Lloyd-Jones, D.M., Allen, N.B., Anderson, C.A.M., Black, T., Brewer, L.C., Foraker, R.E., Grandner, M.A., Lavretsky, H., Perak, A.M., Sharma, G. and Rosamond, W. (2022). Life’s Essential 8: Updating and Enhancing the American Heart Association’s Construct of Cardiovascular Health: a Presidential Advisory from the American Heart Association. Circulation, 146(5). doi:https://doi.org/10.1161/cir.0000000000001078.
[7] Shan, Z., Ma, H., Xie, M., Yan, P., Guo, Y., Bao, W., Rong, Y., Jackson, C.L., Hu, F.B. and Liu, L. (2015). Sleep Duration and Risk of Type 2 Diabetes: A Meta-analysis of Prospective Studies. Diabetes Care, 38(3), pp.529–537. doi:https://doi.org/10.2337/dc14-2073.
[8] Sabia, S., Fayosse, A., Dumurgier, J., van Hees, V.T., Paquet, C., Sommerlad, A., Kivimäki, M., Dugravot, A. and Singh-Manoux, A. (2021). Association of sleep duration in middle and old age with incidence of dementia. Nature Communications, 12(1), p.2289. doi:https://doi.org/10.1038/s41467-021-22354-2.
[9] Hill, E.D., Kashyap, P., Raffanello, E., Wang, Y., Moffitt, T.E., Caspi, A., Engelhard, M. and Posner, J. (2025). Prediction of mental health risk in adolescents. Nature Medicine, 31, pp.1–7. doi:https://doi.org/10.1038/s41591-025-03560-7.
[10] Mei, Z., Cai, C., Luo, S., Zhang, Y., Lam, C. and Luo, S. (2024). The efficacy of cognitive behavioral therapy for insomnia in adolescents: a systematic review and meta-analysis of randomized controlled trials. Frontiers in Public Health, 12. doi:https://doi.org/10.3389/fpubh.2024.1413694.